For example, ATP inhibits phosphofructokinase-1 (PFK1) and pyruvate kinase, two key enzymes in glycolysis, effectively acting as a negative feedback loop to inhibit glucose breakdown when there is sufficient cellular ATP.Ĭonversely, ADP and AMP can activate PFK1 and pyruvate kinase, serving to promote ATP synthesis in times of high-energy demand. The enhancement or inhibition of ATP synthase is a common regulatory mechanism. Many feedback mechanisms are in place to ensure the maintenance of a consistent ATP level in the cell. The routine intracellular concentration of ATP is 1 to 10 uM. ATP must continuously undergo replenishment to fuel the ever-working cell. The process of ATP hydrolysis to ADP is energetically favorable, yielding Gibbs-free energy of -7.3 cal/mol. Through metabolic processes, ATP becomes hydrolyzed into ADP, or further to AMP, and free inorganic phosphate groups. 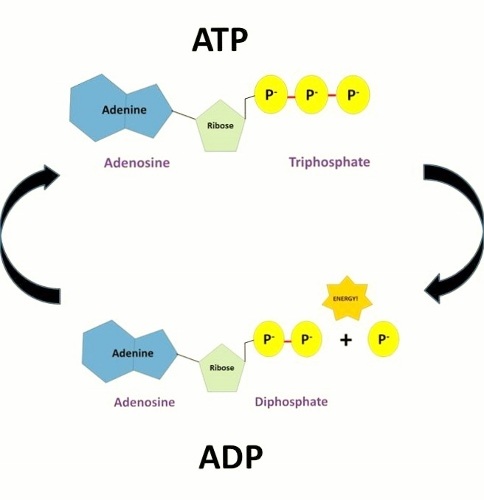
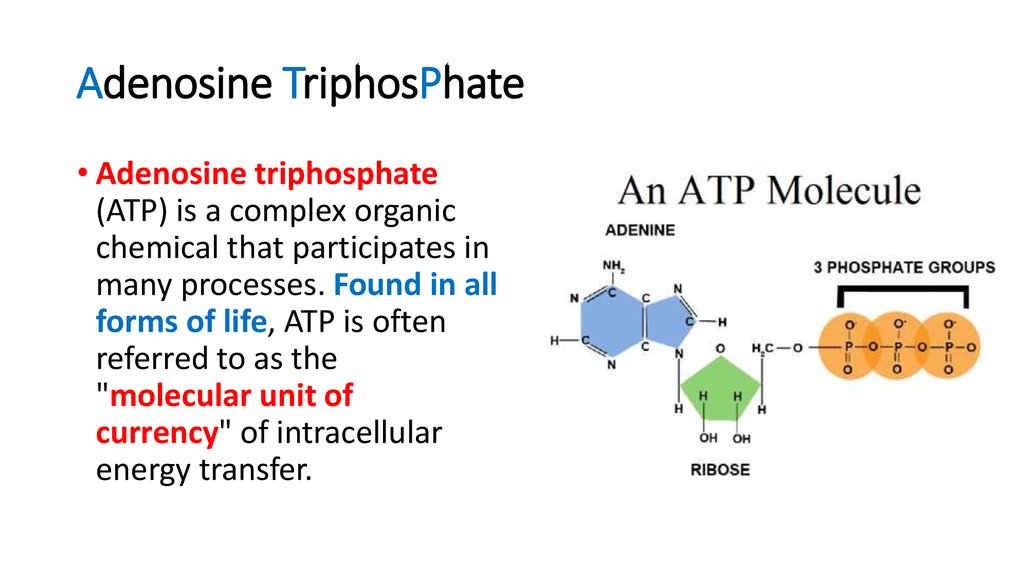
A significant quantity of energy remains stored within the phosphate-phosphate bonds. These bonds are high energy because of the associated electronegative charges exerting a repelling force between the phosphate groups. In the forthcoming sections, ATP will undergo further evaluation of its role as a crucial molecule in the daily functioning of the cell.ĪTP is an excellent energy storage molecule to use as "currency" due to the phosphate groups that link through phosphodiester bonds. As a result, cells within the human body depend upon the hydrolysis of 100 to 150 moles of ATP per day to ensure proper functioning. These processes, as well as others, create a high demand for ATP. ATP is consumed for energy in processes including ion transport, muscle contraction, nerve impulse propagation, substrate phosphorylation, and chemical synthesis. The majority of ATP synthesis occurs in cellular respiration within the mitochondrial matrix: generating approximately thirty-two ATP molecules per molecule of glucose that is oxidized. ATP synthesis utilizes energy obtained from multiple catabolic mechanisms, including cellular respiration, beta-oxidation, and ketosis. In addition to providing energy, the breakdown of ATP through hydrolysis serves a broad range of cell functions, including signaling and DNA/RNA synthesis. The body is a complex organism, and as such, it takes energy to maintain proper functioning. Adenosine triphosphate (ATP) is the source of energy for use and storage at the cellular level. The structure of ATP is a nucleoside triphosphate, consisting of a nitrogenous base (adenine), a ribose sugar, and three serially bonded phosphate groups. ATP is commonly referred to as the "energy currency" of the cell, as it provides readily releasable energy in the bond between the second and third phosphate groups.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
